Description
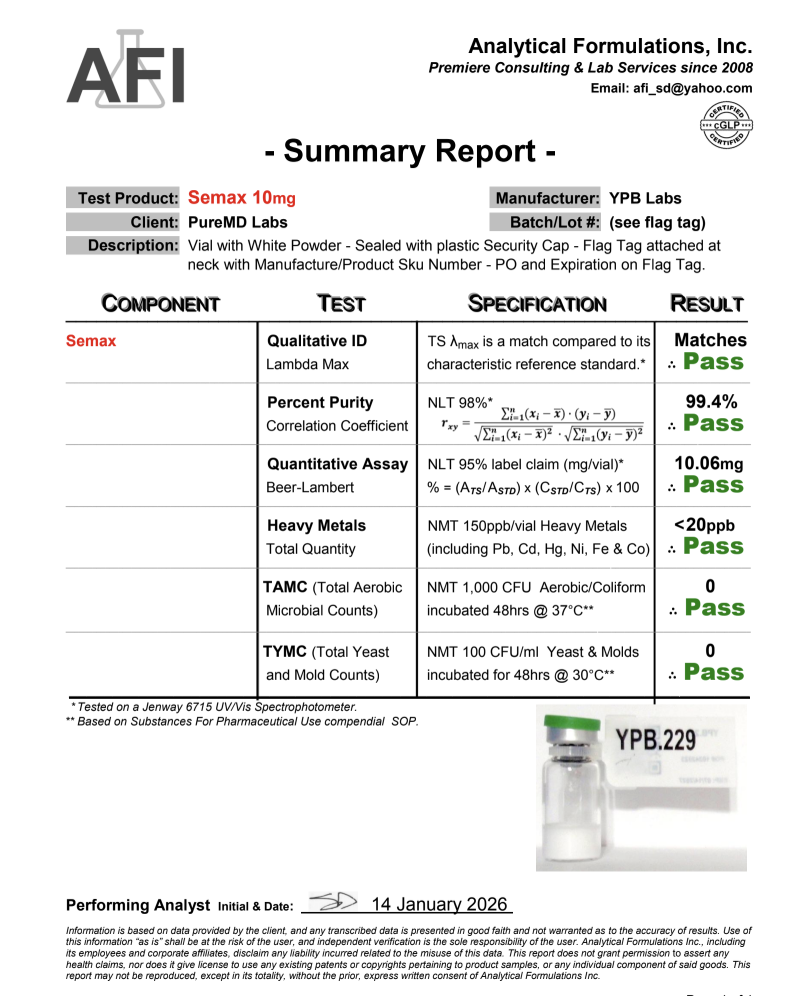
Semax
Synthetic Heptapeptide (ACTH Fragment Analog)
Semax is a synthetic heptapeptide derived from a fragment of adrenocorticotropic hormone (ACTH 4–10), structurally modified to enhance stability in experimental systems. In laboratory and preclinical research contexts, Semax is utilized as a research compound for investigating neuropeptide signaling, synaptic communication, and intracellular signaling pathways associated with neural plasticity.
In experimental models, Semax has been explored for its interactions with monoaminergic systems, neurotrophic signaling pathways, and transcriptional regulation networks. Its short peptide length and defined sequence support controlled studies of peptide–receptor interactions and downstream cellular signaling responses. Semax is often incorporated into research designs examining signal transduction dynamics in neural tissue models.
Rather than functioning as an endogenous hormone, Semax is studied for its neuromodulatory signaling properties, particularly in relation to intracellular cascades and gene expression patterns relevant to neural system research.
Peptide Identity and Molecular Profile
| Property | Description |
|---|---|
| Peptide Name | Semax |
| Peptide Class | Synthetic heptapeptide |
| Structural Origin | Modified ACTH (4–10) fragment |
| Amino Acid Length | 7 residues |
| Peptide Sequence | Met-Glu-His-Phe-Pro-Gly-Pro |
| Molecular Weight | ~813 Da (may vary by synthesis and salt form) |
| Primary Research Focus | Neuropeptide and neural signaling |
Chemical and Registry Information
| Property | Value |
|---|---|
| Molecular Formula | Reported in research literature; may vary by salt form |
| CAS Number | 80714‑61‑0 |
| Registry Identifiers | Research designation: Semax |
| Synonyms | ACTH(4–7)-PGP, Semax peptide |
| Structural Features | Linear peptide; C-terminal Pro-Gly-Pro motif |
Biological Pathways Studied (Preclinical Research)
In laboratory and preclinical research environments, Semax has been studied for its interactions with neural signaling and intracellular regulatory systems. Research emphasizes mechanistic pathways rather than clinical or physiological outcomes.
| Pathway / System | Research Context |
|---|---|
| Monoaminergic Signaling | Studied in dopamine and serotonin pathway models |
| Neurotrophic Factor Signaling | Explored in relation to BDNF-associated pathways |
| MAPK/ERK Pathway | Investigated in intracellular signal transduction |
| Gene Expression Regulation | Examined for transcriptional response patterns |
| Synaptic Signaling Networks | Studied in neural plasticity models |
Research Applications
Semax is commonly used in laboratory research involving:
-
Neuropeptide signaling investigations
-
Synaptic communication and plasticity studies
-
Intracellular signaling pathway analysis
-
Neurotrophic signaling research models
-
Comparative studies of ACTH-derived peptides
All applications are restricted to preclinical, in vitro, or animal research contexts.
Storage and Handling Guidelines
Semax should be stored under controlled laboratory conditions in a cool, dry environment and protected from light. Standard laboratory safety and peptide-handling procedures should be followed to preserve chemical integrity and experimental reproducibility.
Lyophilized Powder
Semax is supplied as a lyophilized powder, produced via freeze-drying to remove residual moisture while maintaining peptide structure and stability. This format supports long-term storage, precise reconstitution, and consistency across experimental protocols.
Shelf Life After Reconstitution
Once reconstituted, Semax is no longer in its lyophilized state, and stability is influenced by buffer composition, temperature, handling frequency, and study duration. In research environments, reconstituted peptide material is generally regarded as suitable for short-term experimental use, with stability considerations incorporated into experimental planning and data-quality assessment. Actual stability may vary depending on laboratory-specific conditions.